/accurate-illustration-of-the-periodic-table-82020791-57cc76f23df78c71b66efbd7.jpg)
In other words, even though an element group might break a trend, the elements within the group display periodic properties.\): Piece of silver. However, the behavior of the noble gases is periodic. The noble gases are an exception to the trend since these elements have filled electron valence shells and electron affinity values approaching zero. group, in chemistry, a column in the periodic table of the chemical elements.In a group, the chemical elements have atoms with identical valence electron counts and identical valence vacancy counts. Nonmetals usually have higher electron affinities than metals. Electron affinity increases moving across a period and decreases moving down a group. Electron Affinity - This is a measure of readily an atom accepts an electron.Electronegativity is an important quantity in determining the nature of bonds between. There is a large difference in electronegativity for atoms from the left- and right-hand sides of the periodic table. Group 2: The alkaline earth metals, such as beryllium, magnesium, and calcium, are also good conductors. 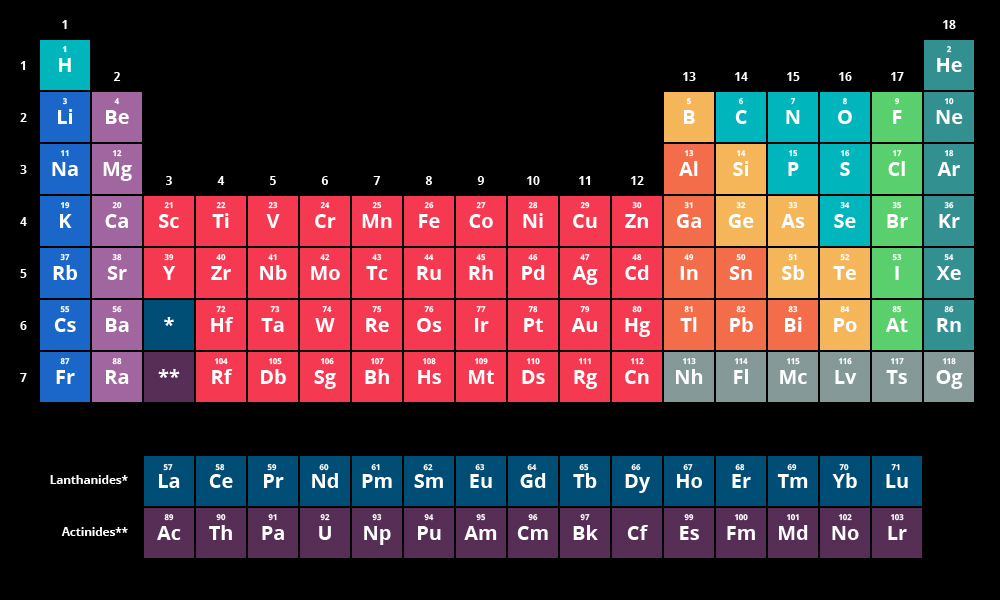
Atom and ion sizes shrink moving across a period because the increasing positive charge of the nucleus pulls in the electron shell. Electronegativity is a chemical property that describes the power of an atom in a molecule to attract shared-pair electrons towards itself. The elements that are generally good conductors of electricity belong to the following groups on the periodic table: Group 1: The alkali metals, such as lithium, sodium, and potassium, are highly conductive. Although it might seem like increasing the number of protons and electrons in an atom would always increase its size, the atom size doesn't increase until a new electron shell is added. Ionic radius is the distance for ions of the atoms and follows the same trend. Atomic radius decreases moving left to right across a period and increases moving down a group. Elements in the periodic table are arranged in periods (rows) and groups (columns). The elements are arranged in seven horizontal rows, called. 1, reacts readily with metals to form compounds that can be broadly classified as salts and, therefore, are known as the halogens, which is derived from a combination of Greek words that translate to 'salt makers. Each box represents an element and contains its atomic number, symbol, average atomic mass, and (sometimes) name. Each of the elements found in this column, which are boxed in lavender in Figure 2.2.1 2.2. Atoms of an atomic number may have varying numbers of neutrons (isotopes) and electrons (ions), yet remain the same chemical element. A modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column (Figure 3.2b). (credit a: modification of work by Serge Lachinov credit b: modification of work by Den fjättrade ankan/Wikimedia Commons) By the twentieth century, it became apparent that the periodic relationship involved atomic numbers. 
Atomic Radius - This is half the distance between the middle of two atoms just touching each other. The periodic table lists elements by atomic number, which is the number of protons in every atom of that element. Figure 2.25 (a) Dimitri Mendeleev is widely credited with creating (b) the first periodic table of the elements. /PeriodicTableWallpaper-56a12a3a3df78cf772680422.jpg)
What Is Ionic Radii How It Varies In Modern Periodic Table Join BYJUS To Explore The World Of Chemistry By Playing With Elements. Atomic No. Periodicity is one of the most fundamental aspects of the periodic table of the elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
